Accelerating next-generation protein engineering
Croucher scholars have led and been involved in a breakthrough to engineer clinically useful proteins such as antibodies and enzymes.
The CRISPR-Cas enzyme is an important tool in medical research for gene editing. To improve its accuracy and efficiency in editing and correcting gene mutations in patients, optimisation of the enzyme’s amino acid sequences is required.
A research team led by Dr Alan Wong Siu-lun (Croucher Fellowship 2012, Butterfield Croucher Studentship 2008) of the Li Ka Shing Faculty of Medicine, University of Hong Kong (HKUMed), has developed the first platform for assembling and barcoding protein-encoding sequences carrying multiple mutations en masse, and has coupled it with next-generation sequencing to track all variants in an unprecedented throughput.
This platform will help researchers to understand epistatic mutations and accelerate next-generation engineering of genome-editing enzymes with new or improved properties.
The research has been published in Nature Methods and a patent application has been filed. Dr Chris Wong Koon-ho (Croucher Fellowship 2009), of the Faculty of Health Sciences, University of Macau, was among the research team members.
The challenge for engineering the CRISPR-Cas enzyme, as well as other proteins, has been to identify the optimal sets of amino acid changes without under- or over-engineering the protein. The combined effect of multiple amino acid changes is difficult to predict, and the number of possible protein-variant combinations increases dramatically with every additional amino acid being modified.
Traditional site-directed mutagenesis techniques for creating variants one-by-one are not feasible for generating a large library of variants with multiple amino acid changes. They are also typically applied in bacterial cells, which limits their utility for engineering genome-editing enzymes and other therapeutic proteins for use in human cells.
The HKUMed platform, named CombiSEAL, can identify new high-fidelity variants of a genome-editing Cas9 protein that have enhanced gene-editing specificity without sacrificing potency and broad targeting range. This high throughput-screening platform aims to lead discovery and optimisation of more clinically useful proteins. The low-cost, easy-to-implement and scalable strategy should help researchers excel in the increasingly competitive environment of innovative bioproducts.
Streptococcus pyogenes Cas9 (SpCas9) nuclease is the most widely used CRISPR-based genome editing enzyme. Using CombiSEAL, a library of 948 SpCas9 variants each of which contains between one and eight mutations, was assembled and delivered into human cells, and their genome-editing efficiency and specificity were systematically quantified. The total number of SpCas9 protein variants characterised in this study greatly exceeds the sum of all other major studies reported thus far.
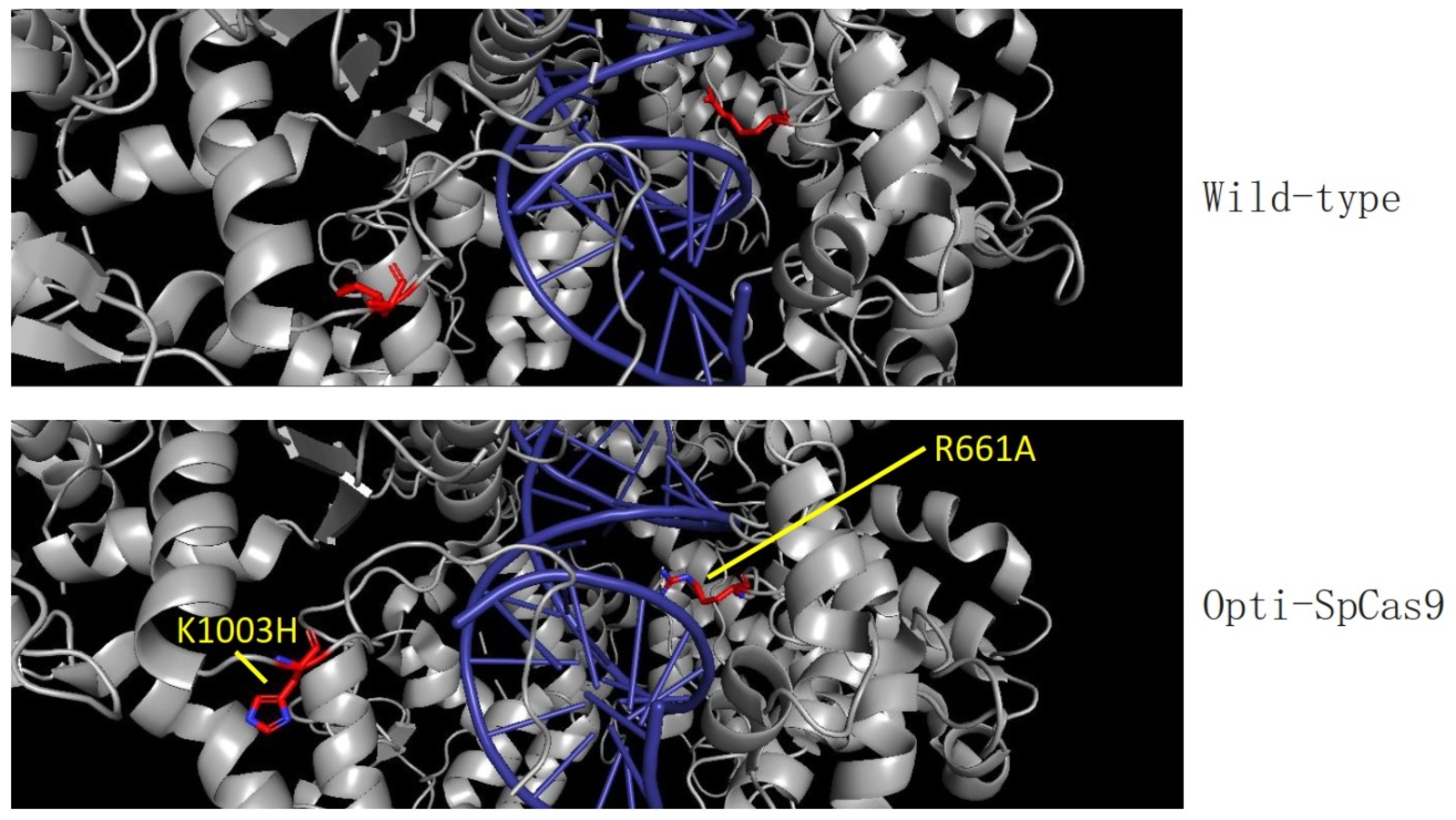
The novel HKUMed strategy will allow the team to scale up the engineering process. The team also identified new high-fidelity SpCas9 variants, Opti-SpCas9 and OptiHF-SpCas9, which generate much fewer undesired off-target genome edits without sacrificing on-target editing efficiency and broad targeting range. CombiSEAL greatly enhances the throughput for functional impact analysis of a massive number of mutation combinations by a simple one-pot reaction library assembly and a round of short-read sequencing of the variant-specific barcode combination.
Dr Alan Wong explained: “This is a large-scale build-and-test platform for combinatorial optimisation of proteins. The one-pot assembly and barcoding strategy substantially reduce time and cost for the whole process, from building to functional characterisation of the entire variant library.”
He added: “Genome-editing tools need to be accurate to maximise safety for clinical use, while high editing efficiency and a broad target range are also important for applications such as CRISPR screens.”

Dr Alan Siu-lun Wong is an Assistant Professor at the University of Hong Kong, where he holds a joint appointment at the School of Biomedical Sciences and Department of Electrical and Electronic Engineering. He obtained his BSc and MPhil degrees in Biochemistry and Molecular Biotechnology from the Chinese University of Hong Kong in 2005 and 2007 respectively, and completed his PhD in Biochemistry at the Hong Kong University of Science and Technology in 2011. He undertook postdoctoral training with the Synthetic Biology Group at Massachusetts Institute of Technology from 2012-2016 before joining HKU. Alan received a Croucher Fellowship in 2012 and a Butterfield Croucher Studentship in 2008.
To view Dr Alan Wong’s Croucher profile, please click here.

Dr Chris Wong Koon Ho is an Assistant Professor in the Faculty of Health Sciences, University of Macau. He received his BSc (Hons) and PhD (Genetics) degrees from the University of Melbourne, Australia, and moved to Harvard Medical School for his post-doctoral research in 2009. He joined University of Macau in 2013. Chris received a Croucher Fellowship in 2008.
To view Dr Chris Wong’s Croucher profile, please click here.