Paving the way for plant-based vaccine technology
Using biological cells as molecular farms that produce proteins to human specifications plays a large and growing role in medicine since the 1980s. So far, mammalian and bacterial cell cultures are the most widely used expression systems.
A well established method to produce, for example, antibodies consists in using Chinese Hamster Ovary cell cultures, while E. Coli bacteria are harnessed to produce insulin for human use.
For conventional cell cultures of this type, there is a long history of established procedures and regulatory frameworks.
This is less the case for plant made pharmaceuticals. As a consequence, they currently play a lesser role in the market for biopharmaceuticals overall.
Plant made pharmaceuticals however, have great potential – especially when it comes to the production of pharmaceuticals that are needed quickly, at low cost and at great scale. A case in point: Vaccines that are needed for a very large number of people.
A recent study published by Frontiers in Plant Science stated that in future, “the production of vaccines, therapeutics, and diagnostics in plants has the potential to function as a first line of defence against pandemics.“
To a limited extent, this is already happening. For example, in February 2022, Canadian regulators approved a plant-based COVID 19 vaccine.
More recently, two Hong Kong scientists contributed to further paving the way for plant-based vaccines.
In a paper published in Proceedings of the National Academy of Sciences of the USA, Professor Wong Kam Bo (Croucher Scholarship 1993) and Professor Jiang Liwen (Croucher Senior Research Fellowship 2009 and 2015), both professors of life science at Chinese University of Hong Kong, discovered a key mechanism behind storage protein accumulation in plant seeds.
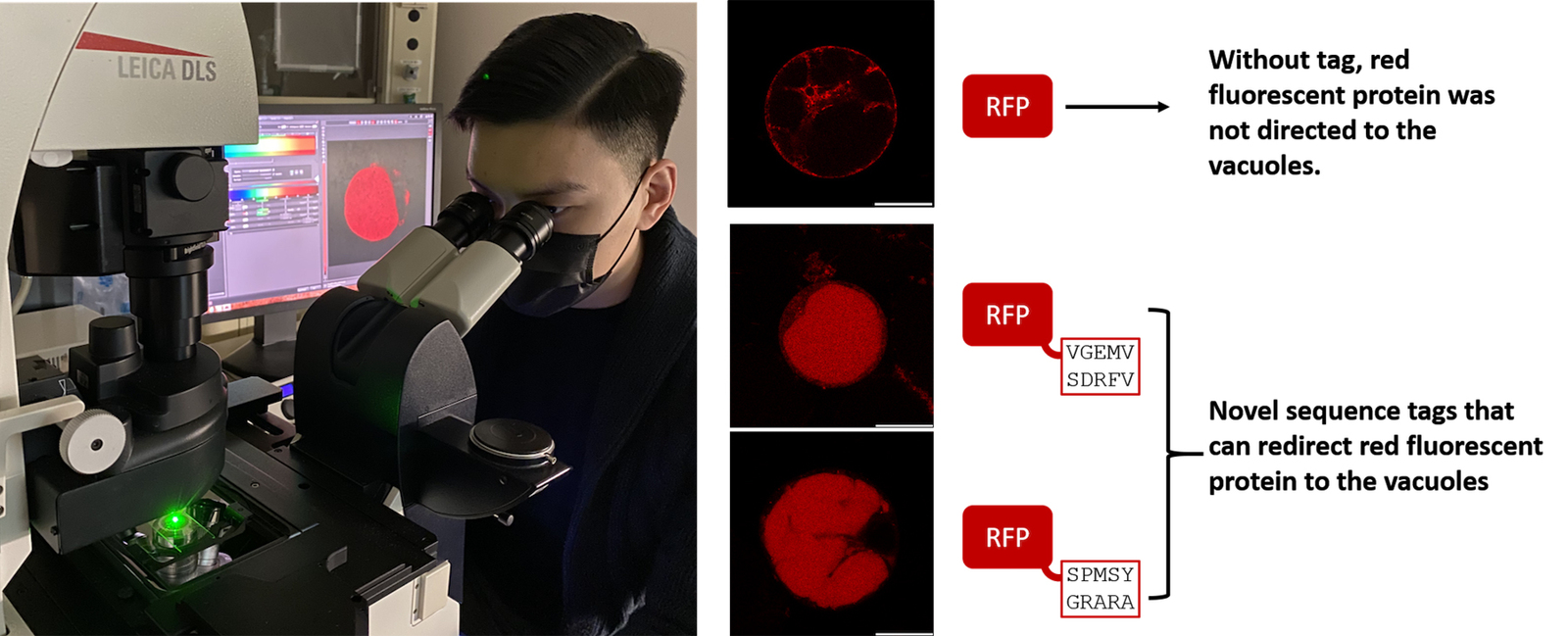
“Our research shows for the first time that, in principle, we can redirect proteins to go to the vacuoles in plant cells“, says Wong. The team plans to apply the research results to engineer proteins for expression and storage in plant seeds.
Game changer
“This is an important finding“, Wong adds. “It opens up the possibility of seed storage protein manipulation. We would be able to direct biotechnologically important proteins such as antigens for vaccine production to the vacuoles.”
This would transform the plant into a bioreactor to produce and store millions of copies of the proteins in question.
“This could be a game changer for various biotechnological applications”, says Wong.

Different strategies for plant made pharmaceuticals
There are several strategies to “express” or produce proteins in plant cells.
One such approach is to use transgenic plant cell cultures. This is currently in use for the production of a drug for the treatment of Type 1 Gaucher’s disease.
Another strategy is to express the proteins in the leaves of crop plants. A recent example for this is utilising tobacco leaves to produce antibodies that can treat the Ebola virus.
Wong and Jiang have focused on a third approach: To express and store proteins in the seeds of edible plants.
Edible vaccines?
Seeds have a number of advantages over other expression systems.
“Seeds are the natural place to store proteins in plants“, says Jiang. “Proteins stored there are more stable and do not suffer a loss of activity during storage.“
This is especially relevant for drugs that are to be distributed on a mass scale, such as vaccines. Drugs that require, for example, sophisticated cold-chains to keep them stable and effective are inherently difficult to distribute. This can limit their use to countries where such high logistical demands can be met.
In addition to ease of storage and therefore distribution, seeds as expression systems can also maximise the output. “Seeds make it easier to scale up production of the desired proteins and to grow them in a locally concentrated fashion, rather than distributing them all over the plant”, says Jiang.
The expression of plant-based pharmaceuticals in edible seeds also brings another advantage: ”It can allow the administration of these pharmaceuticals via the oral route, for instance as edible vaccines”, says Wong.

How to tell a plant what it should do
Focusing on seeds as an expression system introduces a new problem, however: In addition to prompting the plant to grow the protein you want to harvest, you now also have to tell that plant where exactly to store that protein.
Is it possible to direct a plant to do this?
This is what Wong and Jiang have shown to be possible. They have recently discovered the mechanism by which a plant recognises and transports its seed proteins to the final destination.
In addition, they have designed a way to redirect biotechnically important proteins to exactly where they need to go.
Bar codes written in amino acids
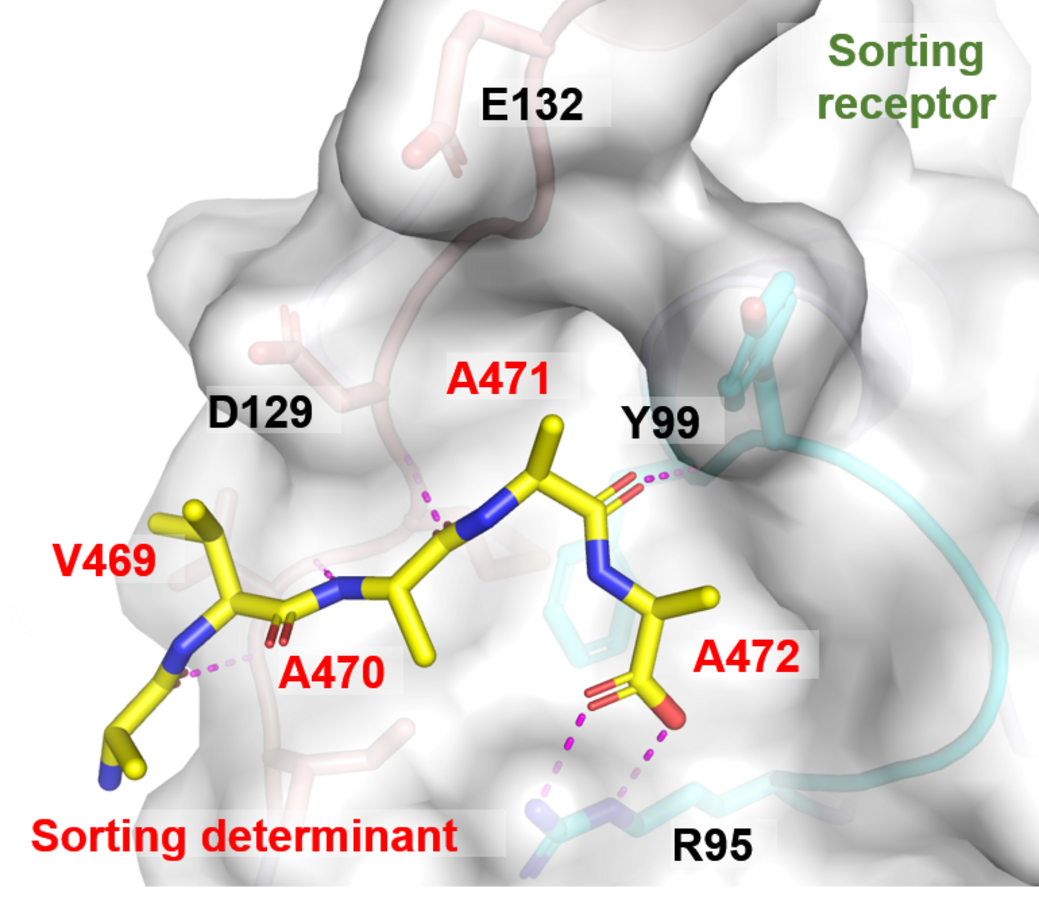
“The question we addressed is: By what mechanism are proteins within the seed selected to be stored in particular compartments in that seed”, says Wong.
“This works rather like a bar code and a bar code reader”, he adds. “There is a certain tag or “bar code” on a given “cargo”, in this case a protein.”
And there is a “bar code reader” within the organism that makes sense of this tag. This determines, within the plant, where the cargo needs to go: For instance, to which sub-cellular compartment within the seed.
“Storage proteins contain such a ‘bar code’ in their amino acid sequence”, says Wong. “The vacuolar sorting receptor is a protein that acts as a bar code reader in plant cells. It recognizes the sequence label and carries the cargo proteins to their final destination.”
The team also designed a way to redirect biotechnically important proteins to exactly where they need to go.
They used X-ray crystallography as a molecular microscope to visualise how the sequence label on a storage protein is recognised and succeeded in redirecting a fluorescent protein by tagging the protein with a sorting determinant – a new “bar code.”
This would imply it is possible to direct a desired “cargo” – for instance a medically useful protein – to exactly those sub-cellular compartments that maximise the benefit of those proteins for human use.
The compartments in question are called protein storage vacuoles in seeds. This is where the plant naturally stores its own proteins to facilitate the growth of the seedlings during the early stages of seed germination.
It is also an ideal place for scientists and technicians to store the proteins they prompted the plant to produce on their behalf – for example medically useful proteins such as antigens for vaccine production.
Although their research is yet for direct application, it is an important proof of concept given the potential of plant made pharamceuticals for the production of vaccines, therapeutics and diagnostics.
Given the potential of plant made pharmaceuticals for the production of vaccines, therapeutics, and diagnostics, however, this is an important proof of concept.
“In principle”, says Wong, “all proteins of value could be produced by or could benefit from using this strategy.“